Typical Materials Used in GES Batteries and Supercapacitors
NanoLab, Inc. Buckypaper* SEM 30K magnification Wikipedia – CNT *Nano-Lab, Inc., Web at, http://www.nano-lab.com/buckypaper.htm - Electrical properties
Cohesive nonwoven CNT on the left is alongside a 30,000 magnification in the middle. At super magnification strands look like a Wikipedia picture on the right. GES’s 3-D nanoscale architecture uses these materials or similar graphene forms as sheets. These are typical materials used in GES ultra-low resistance batteries and supercapacitors.
Cohesive nonwoven CNT on the left is alongside a 30,000 magnification in the middle. At super magnification strands look like a Wikipedia picture on the right. GES’s 3-D nanoscale architecture uses these materials or similar graphene forms as sheets. These are typical materials used in GES ultra-low resistance batteries and supercapacitors.
Low Resistance Attachment of Materials to Metal
The nanoscale graphene materials containing lodged and immobilized faradaic materials as pictured above are electrically attached to current collector metal backing by strong, closely spaced, multi-point attachment comprising electrical conduction sinks. This produces ultra-low electrical resistance between the electrochemistry and the metal. The plurality of electrical conduction sinks uses 5% or less of total electrode area.
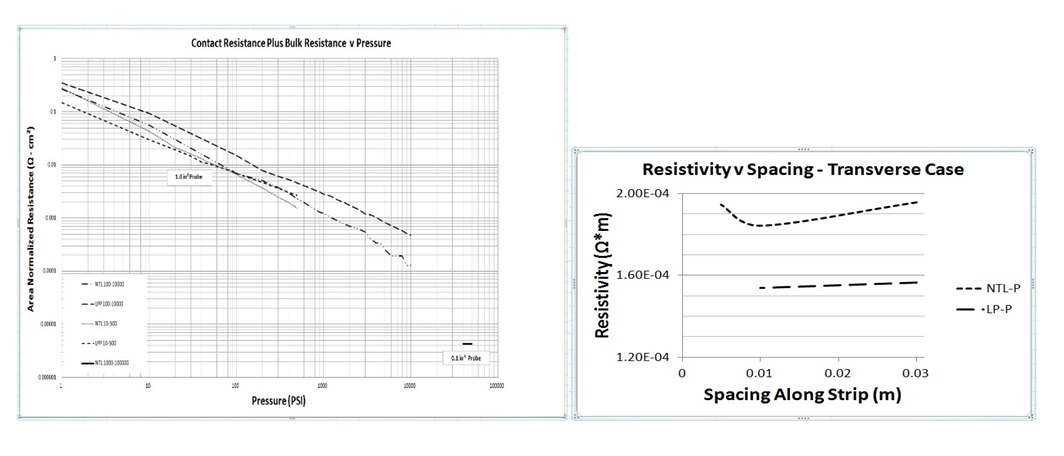
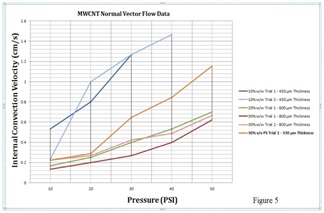
Electrode Impedance Measurements Proof of Concept
GES experimental resistance data proves the concept.
Battery Materials Lodged and Immobilized Within Electrode Structure
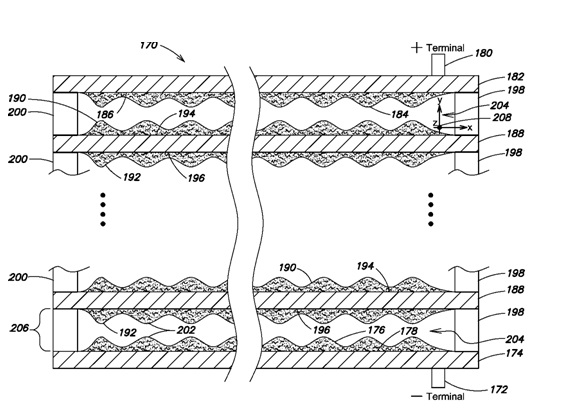
3-D Nanoscale High Voltage/Power Architecture
GES 3-D nanoscale battery and super-capacitor architecture provides maximum material energy density using any electrochemical cell chemistry known in the art. The above illustrates an arbitrarily high voltage cell with as many unit cells in the stack as required to achieve unit cell voltage multiplication. The combination of high current density and high voltage provides compatibility with unprecedented charging power. Recharge in minutes instead of hours is now possible. Metal sheet and attached electrode thicknesses are selected to optimize ion kinetics.